HealthRise program
We conducted process and endline evaluation of access to chronic care for hypertension and diabetes in underserved communities in Brazil, India, South Africa, and the United States. HealthRise was a five-year global program funded by the Medtronic Foundation, designed to improve access to chronic care for individuals in underserved communities suffering from hypertension and diabetes. Its primary objectives were to increase screening, diagnosis, management, and control of cardiovascular disease and diabetes. The program was implemented in nine communities in Brazil (2 sites), India (2 sites), South Africa (2 sites), and the United States (3 sites) and grantees in each location developed programs uniquely tailored to each local context. Despite substantial variation in program design, all shared the same core principles: empowering individuals living with cardiovascular disease and diabetes, enabling frontline health workers to better address the needs of these individuals and communities, and supporting advocacy and policy that promotes effective care for non-communicable diseases.
HealthRise was a five-year global program funded by the Medtronic Foundation, designed to improve access to chronic care for individuals in underserved communities suffering from hypertension and diabetes. Its primary objectives were to increase screening, diagnosis, management, and control of cardiovascular disease and diabetes. The program was implemented in nine communities in Brazil (2 sites), India (2 sites), South Africa (2 sites), and the United States (3 sites) and grantees in each location developed programs uniquely tailored to each local context. Despite substantial variation in program design, all shared the same core principles: empowering individuals living with cardiovascular disease and diabetes, enabling frontline health workers to better address the needs of these individuals and communities, and supporting advocacy and policy that promotes effective care for non-communicable diseases.
IHME served as the independent evaluation partner and carried out three primary activities:
- Establishing baseline measurements and conducting a needs assessment
- Managing program monitoring
- Conducting an endline evaluation
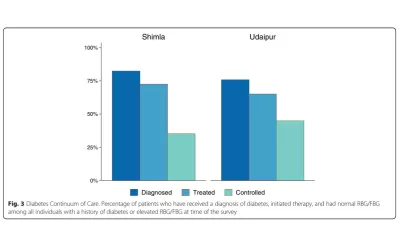
The global framework for needs assessment and monitoring and evaluation was designed to align with Medtronic Foundation’s Continuum of Care.
The specific objectives of the needs assessments were to:
- Estimate the prevalence of diseases and risk factors
- Identify major gaps along the continuum of care
- Identify supply- and demand-side barriers that contribute to these gaps
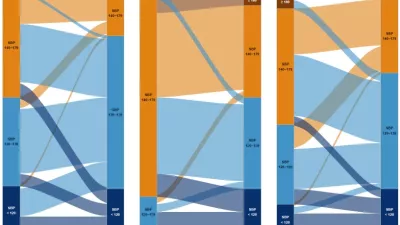
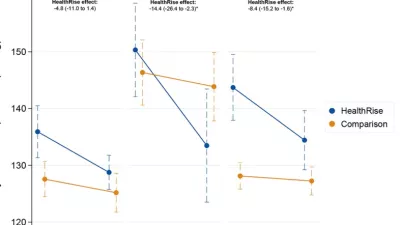
The monitoring and evaluation activities conducted by IHME can be broadly grouped into two categories: the process evaluation and the endline evaluation. The process evaluation, which had substantial overlap with the monitoring phase, offered insights into the various activities being executed across each of the implementation programs and the endline evaluation measured changes in target health outcomes. Key outcome indicators for HealthRise included the number of individuals diagnosed with diabetes with their most recent blood glucose measurement below the threshold for controlled disease and the number of individuals diagnosed with hypertension with a decrease of 10% or more between their first and last blood pressure measurement.
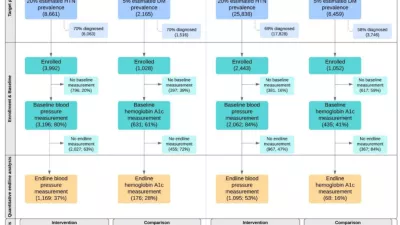
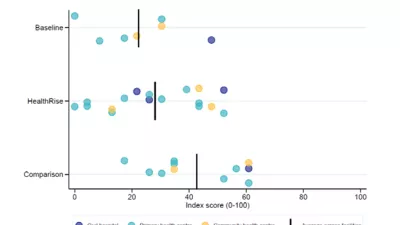
For the endline evaluation, a mixed methods quasi-experimental approach was implemented in which both quantitative and qualitative data captured the different types of needs, barriers and opportunities. A combination of quantitative and/or qualitative data were collected at each site, and when possible, data were collected from both intervention and comparison areas. These data were compared with data collected at baseline to assess changes in patient outcomes, knowledge, and behaviors; provider knowledge and practices; the quality and availability of health services; and the reach of HealthRise programs.
Survey instruments and protocols
Quantitative survey instruments are comprised of a health facility questionnaire and patient exit interviews including the collection of biomarkers. IHME developed and programmed all quantitative survey instruments in Surveybe, a computer-assisted personal interview data collection platform. Local data collection teams administered surveys on tablets in India and South Africa. Quantitative surveys were not conducted in Brazil or the US. All quantitative survey instruments and collected data are published and publicly available on IHME’s Global Health Data Exchange.
Qualitative data was collected in the form of focus group discussions and key-informant interviews. All interview guides and protocols were developed by IHME. Local data partners conducted qualitative data collection in Brazil, India, and South Africa. IHME conducted qualitative data collection in the US.
Data Collection Tools
Brazil
- Endline Qualitative Instruments
- Brazil, Interview Guide and Protocol
India
- Endline Qualitative Instruments
- India, Interview Guide and Protocol
- Endline Quantitative Survey Instruments*
South Africa
- Endline Qualitative Instruments
- Pixley ka Seme, Interview Guide and Protocol
- uMgungundlovu, Interview Guide and Protocol
- Endline Quantitative Survey Instruments*
US
- Endline Qualitative Instruments
- Hennepin County, Interview Guide and Protocol
- Ramsey County, Interview Guide and Protocol
- Rice County, Interview Guide and Protocol
*Note: All “Patient Survey” and “Facility Survey” data collection was conducted electronically via tablets using SurveyBe CAPI software. The guides are intended to provide readers with documentation of survey content, and were not used to collect data in the form in which they are presented here.
Funding and partners
The program was funded by Medtronic Foundation. The HealthRise Evaluation team collaborated with and harnessed the expertise of numerous distinguished organizations to carry out this work:
Global Team
- Medtronic Foundation
- Abt Associates
- Human Sciences Research Council (HSRC)
- Instituto Sírio-Libanês de Ensino e Pesquisa (IEP)
Grantee Partners
- HealthFinders Collaborative, Inc (HFC)
- Regions Hospital Foundation
- Pillsbury United Communities (PUC)
- FAPEX
- FUNDEP
- Project HOPE
- Expectra Health Solutions
- Catholic Health Association (CHAI)
- MAMTA Health Institute for Mother and Child (MAMTA)
Data Collection Partners
- Dinamica Cursos
- Tesla Gestao
- Press Consultoria
- Indira Gandhi Medical College (IGMC) Shimla
- GfK Mode (Growth from Knowledge)
- Development Solutions, Inc
- Social Surveys Africa